The Power of Latent Heat
Water comes in three forms: liquid (water), gas (water vapor) and solid (ice). We are familiar with the process of converting ice to water, known as melting or fusion. Not only does melting require temperatures to warm or energy, but there is also energy required to physically change the form of the H20. Similarly energy is required or released when changing between the other phases. Since this energy is not readily realized, it is referred to as latent, or hidden, energy.
The specific heat of water is 1, which means that it takes 1 calorie to heat up 1 gram of water by 1 degree Celsius. How much energy do you think it takes to melt 1 gram of ice, raise it's temperature by 100C and vaporize it? 720 calories. However, only 100 calories to take water at 0C and warm it up to the boiling point of 100C. The rest of that energy is latent heat!
Let's consider the migration of water from the Earth's surface into the atmosphere. Surface water is evaporated requiring energy that would otherwise be spent to heat up surface temperature. Air temperatures in regions with wet surfaces that undergo significant evaporation, generally stay much cooler during the daytime hours (when evaporation peaks), that comparable dry surfaces. This latent heat of evaporation is often thought of as a cooling process; however, it most accurately reflects a weaker amount of warming than would otherwise occur. This effectively removes energy from the Earth's surface and puts it into the atmosphere as water vapor. This water vapor eventually rises and or cools to its dewpoint at which point condensation occurs converting the water vapor back into its liquid form as a cloud droplet. This process releases heat to the atmosphere. Effectively, surface evaporation and condensation of cloud droplets transfers energy from the surface to the atmosphere.
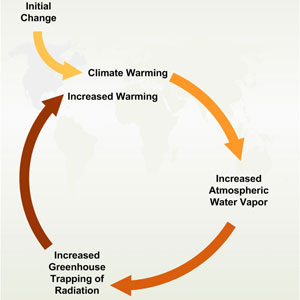
Let's go back to climate change -- we said that as the amount of carbon dioxide in the atmosphere increases, it traps more heat energy and warms the atmosphere and the surface. With more energy available at the surface (especially over the 70% of the Earth surface that is oceans), there is more evaporation, and more water vapor is added to the atmosphere. Since the air temperature increases, the air can effectively hold more water vapor before condensation occurs. The increase in total water vapor in the atmosphere strengthens the greenhouse effect and serves to warm the atmosphere even more and cause more evaporation. This positive feedback loop is called the water vapor feedback loop.
The specific heat of water is 1, which means that it takes 1 calorie to heat up 1 gram of water by 1 degree Celsius. How much energy do you think it takes to melt 1 gram of ice, raise it's temperature by 100C and vaporize it? 720 calories. However, only 100 calories to take water at 0C and warm it up to the boiling point of 100C. The rest of that energy is latent heat!
Let's consider the migration of water from the Earth's surface into the atmosphere. Surface water is evaporated requiring energy that would otherwise be spent to heat up surface temperature. Air temperatures in regions with wet surfaces that undergo significant evaporation, generally stay much cooler during the daytime hours (when evaporation peaks), that comparable dry surfaces. This latent heat of evaporation is often thought of as a cooling process; however, it most accurately reflects a weaker amount of warming than would otherwise occur. This effectively removes energy from the Earth's surface and puts it into the atmosphere as water vapor. This water vapor eventually rises and or cools to its dewpoint at which point condensation occurs converting the water vapor back into its liquid form as a cloud droplet. This process releases heat to the atmosphere. Effectively, surface evaporation and condensation of cloud droplets transfers energy from the surface to the atmosphere.
Let's go back to climate change -- we said that as the amount of carbon dioxide in the atmosphere increases, it traps more heat energy and warms the atmosphere and the surface. With more energy available at the surface (especially over the 70% of the Earth surface that is oceans), there is more evaporation, and more water vapor is added to the atmosphere. Since the air temperature increases, the air can effectively hold more water vapor before condensation occurs. The increase in total water vapor in the atmosphere strengthens the greenhouse effect and serves to warm the atmosphere even more and cause more evaporation. This positive feedback loop is called the water vapor feedback loop.
Log into BBLearn and under Module 4, complete "Assignment 4.2: Phase Changes and Latent Heat Energy."